TRIAL CLOSED
Please note that the trial has now closed.
Summary of the findings of the PIROUETTE trial
The PIROUETTE trial included patients with heart failure with normal forward pumping function of the heart (‘heart failure with preserved ejection fraction’, or ‘HFpEF’ for short), and heart muscle scarring (fibrosis).
Patients underwent a heart MRI scan at the beginning of the study. Patients with evidence of heart muscle scarring, as indicated by a heart MRI measurement called ‘extracellular volume’, were randomly assigned to take pirfenidone or a placebo daily for 1 year. 94 patients were randomised: 47 took pirfenidone and 47 took placebo. After a year, patients underwent a second heart MRI scan to measure the change in heart muscle scarring.
The results showed that pirfenidone reduced heart muscle scarring. Extracellular volume declined by 1.21% more on average in patients who took pirfenidone compared with those receiving placebo.
Fluid retention, measured using a blood test called NT-proBNP, also improved in patients taking pirfenidone compared to those receiving placebo. There were no differences in measurements of heart muscle relaxation.
The most common side effects in patients taking pirfenidone were nausea, insomnia and rash, which are similar to those which lung patients can experience when taking pirfenidone.
The findings suggest that pirfenidone could have favourable effects in patients with HFpEF. Larger trials are now needed to determine whether pirfenidone improves the expected course of HFpEF (prognosis), for example, whether it reduces admissions to hospital for heart failure and heart failure-related deaths.
If you would like to read more about the results, please go to https://www.nature.com/articles/s41591-021-01452-0
The study team would like to thank the patients who took part in PIROUETTE, and their families and carers, and the patients who were part of the study oversight committees. The study was only possible with your help and support.
Overview
PIROUETTE - A double-blind randomised controlled trial of Pirfenidone in patients with heart failure and preserved ejection fraction
This clinical trial has been designed to test whether a drug called pirfenidone leads to a reduction in heart muscle scarring and improves heart function in patients with heart failure.
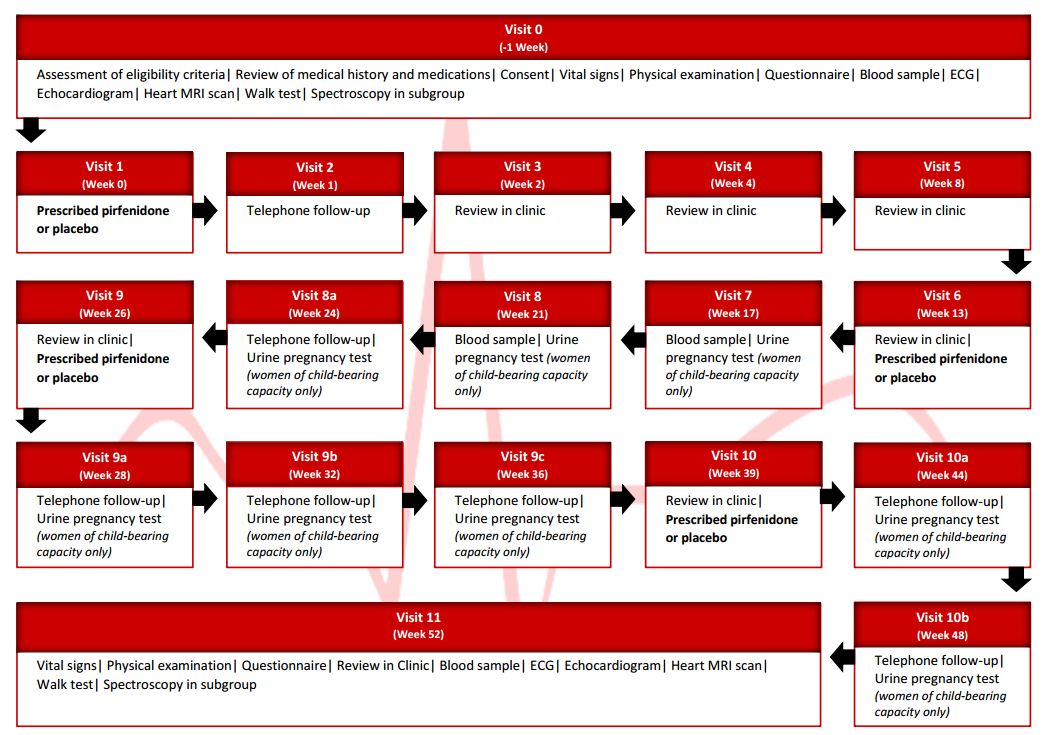
Taking part involves 11 visits to the hospital and 1 telephone call, additional telephone calls will apply to women of childbearing potential to assess home pregnancy testing.
Study tests include: spectroscopy, blood tests, heart trace (ECG), echo (ultrasound scan of your heart), heart magnetic resonance imaging (MRI scan of your heart), short questionnaire, walk test, and pregnancy test if you are female and of childbearing age.